Putting a new spin on immunoassays for cell and gene therapy – Part 2
Careful platform choice generates data with higher quality to support DOE
Harnessing the power of cell and gene therapy involves a large range of analytical methods that can deliver robust results to support rapid data driven decision-making. One assay development team has used immunoassays to track a transgene IgG product. Their choice of immunoassay platform enabled them to generate high quality data that supported Design of Experiments to quickly optimize cell culture.
The power of mRNA-driven therapeutics
AstraZeneca, along with its global biologics research and development unit MedImmune, is making new inroads into gene therapy, especially in respiratory disease. For example, one collaboration involves adeno-associated virus (AAV) gene therapy, vector discovery and product development for the treatment of chronic lung disease. Another is focused on developing new stabilized non-immunogenic modified RNA therapies for respiratory diseases. The transient nature of mRNA, which is situated in the cytoplasm brings flexibility and broad utility to the design of therapeutics that do not require transport across the nuclear membrane.
Using immunoassays to track transgene products
One ongoing project at MedImmune required the quantification of transgene potency of mRNA encapsulated in lipid nanoparticles (mRNA:LNP). Lipid nanoparticles (LNP) are the leading delivery system for small interfering RNA (siRNA) and mRNA for systemic applications. In the case of gene therapy products involving mRNA:LNP constructs, defining identity according to regulatory guidelines involves potency assays (1) to determine potency (transgene expression), mRNA translation, and a transgene identity assay.
The formulation of mRNA:LNP is complex and translation must be checked using a transgene potency assay for release. Assays must be robust, reproducible, quantitative and capable of being validated, and in line with regulatory expectations (including USP 1032, USP 1033, USP 1034, and ICHQ2R guidelines).
The transgene in question encodes an IgG, which meant that an immunoassay was the obvious approach to determining potency. This potency assay is designed to quantify IgG expression to determine biological activity of the mRNA:LNP product. This is different to a typical potency assay for monoclonal antibodies, where the focus is on measurement of biological activity of the antibody products. The bioassay team took a long hard look at the analytical needs and decided that assay range and sensitivity were key parameters due to the limited amounts of mRNA:LNPs that can be delivered to cells due to toxicity at high concentrations. They started to look for immunoassay alternatives, and decided to evaluate Gyrolab® system.
Broadening analytical range, increasing sensitivity, and improving precision to support DOE
Working with bioassay development at MedImmune (AstraZeneca), Uchechukwu (Uche) Okorji-Obike presented (European Gyrolab User Meeting held in Milan in June 2019) how she and her team compared a Gyrolab IgG assay (Fig. 1) with a commercially available ELISA kit in the analysis of the IgG product from the mRNA:LNP batch.
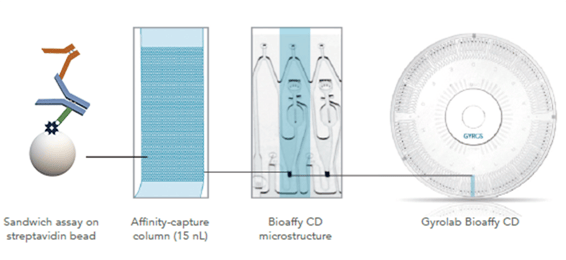
Figure 1. The assay design for Gyrolab IgG Kit is based on the biotinylated Fz fragment as capturing molecule, which is specific for the Fc portion of human intact IgG. An anti-F(ab’)2 antibody is used as detection antibody.
The team at MedImmune was able to quickly establish the Gyrolab assay to measure transgene expression as a measure of potency. Gyrolab system delivered data with higher precision and over a broader dynamic range compared with ELISA, and did not demand the optimization of minimal required dilution (MRD). The higher precision of the Gyrolab assay made it more powerful when applying Design of Experiments (DOE) to method development, for example in analyzing and optimizing the many assay variables, such as cell seeding density, media composition, and incubation time (Fig. 2).
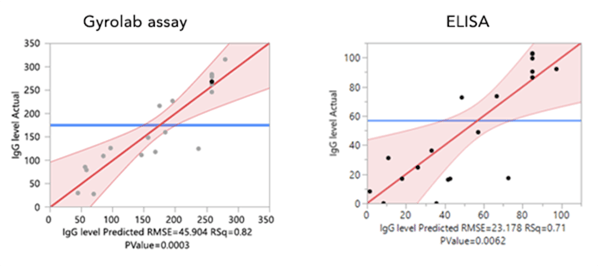
Figure 2. Residual plots from Design of Experiments to maximize IgG production in cell culture based on three factors: cell number, concentration of Fetal Bovine Serum, and amount of mRNA:LNP. The Gyrolab assay gave a much more precise model of actual vs. predicted IgG level than ELISA, which improved the power of the DOE.
The future of Gyrolab system in cell and gene therapy applications
This application, together with the monitoring of lentiviral vector immunogenicity presented in the previous article in this series, demonstrates the real value of Gyrolab systems in supporting the development and evaluation of cell and gene therapy products. While the transgene product studied by MedImmune was an IgG, the application of immunoassays such as Gyrolab assays is not necessarily limited to gene products that result in IgG synthesis. Immunoassays could be used for other gene products providing they can be detected using antibodies. These are just two examples of how Gyrolab system is finding more and more applications in cell and gene therapy, thanks to the ingenuity of assay developers the world over.
You can find out more about Gyrolab IgG Kit (separate kits for low titer and high titer with different dynamic ranges so the scientist can choose the one that is most suitable) by downloading the Product Information Sheet.
References: