Anti-interferon autoantibodies play a major role in the age-related severity of COVID-19

An enormous research effort continues to unravel the complexities of COVID-19. Two major studies published recently based on a massive international collaborative effort led by Rockefeller University in New York City and Institut Imagine in Paris indicate that genetic abnormalities together with autoantibodies that reduce type 1 interferon levels can explain nearly 25% of severe cases of COVID-19. These discoveries, accelerated using automated, microfluidic immunoassays to process over 30,000 samples, may have major consequences on how we handle patient screening and care during the ongoing pandemic.
The excessive impact of COVID-19 on the older population
The COVID-19 pandemic has presented enormous challenges in understanding variations in susceptibility to infection, diversity of symptoms, and severity of disease in patients infected with SARS-CoV-2. One fact became very clear early in the COVID-19 pandemic — the disease was much more serious in the older segment of the population, while young people seemed to be much less affected (Figure 1). A series of recent publications represent a breakthrough in explaining this age-related susceptibility to the disease: it’s all about cytokines.
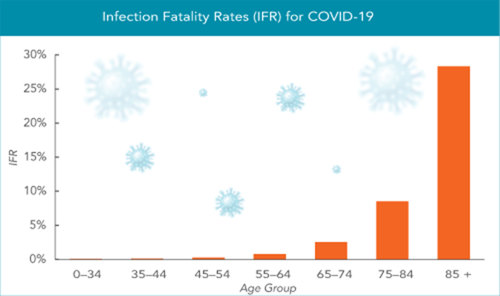
Figure 1. The infection fatality rate (IFR) for COVID-19 increased exponentially with age. IFR: total number of deaths from the disease divided by the total number of infected individuals. Based on data from Levin et al, 2020 (1).
Cytokines and COVID-19
Cytokines are first responders to any pathogen infection including SARS-CoV-2, and it is critical that the resulting immune response matches the pathogen threat — too much and the immune response becomes self-destructive; too little and the defense is insufficient. Both are an issue in COVID-19.
The overzealous reaction of the body to SARS-CoV-2 infection may result in a potentially deadly “cytokine storm” or cytokine release syndrome (CRS) causing an out-of-control inflammatory response. This phenomenon had been seen earlier in other infections such as SARS-CoV-1 and MERS-CoV and is suspected to be the main cause of death in the 1918 Spanish Flu pandemic. The cytokine storm seen in severe COVID-19 is now routinely dampened by low doses of dexamethasone, a steroid drug discovered 60 years ago. In contrast, boosting cytokine levels at least locally can improve the immune response, for example by administering interferon beta to the lungs — a potential treatment that is currently in Phase 3 trials.
One of the top-10 discoveries of 2020
While the cytokine storm is clearly a contributing factor to COVID-19 morbidity, the role of low cytokine levels, especially in the older population, was put into sharp focus in August 2020. A multi-site research group led by immunologist Dr Jean-Laurent Casanova at the Rockefeller University in New York City had shown that 10% of people with severe COVID-19 had autoantibodies against type 1 Interferon (IFN 1; ref 2). This report followed another that described how male-dominated loss-of-function genetic abnormalities in the TLR7 gene blocking IFN 1 production can explain 1.3% of severe forms of COVID-19 (3). Both autoantibodies and the mutation would reduce active IFN 1 levels and handicap the immune response.
These groundbreaking findings linking IFN 1 levels with COVID-19 disease severity were described by Aaron Ring, MD, PhD, an immunologist at the Yale School of Medicine in New Haven, Connecticut, as, “probably one of the most important papers in the pandemic” (4). And the interest in this work continues, as Paul Bastard, MD-PhD, first author of one the publications points out: “The publications resulting from the collaboration between Rockefeller University and Institut Imagine have been very well received, with 50,000 downloads of the latest article published only a month ago”. Their studies published last year were also ranked among the 10 major advances of 2020, according to the prestigious journal Nature (5).
A large-scale study demonstrates the prevalence of anti-type 1 IFN autoantibodies
The evidence for the role of autoantibodies in COVID-19 disease severity was confirmed by a massive follow-up study including the analysis of over 30,000 serum or plasma samples published in August 2021 (6). Key to this study was the use of Gyrolab system to measure anti-type 1 IFN autoantibody levels in COVID-19 patients and healthy individuals.
Ramping up throughput with Gyrolab system
Medetia Pharmaceuticals played a key role in helping Paul Bastard and his colleagues to increase throughput in the follow-up study. The company was founded in early 2019 by Jean-Philippe Annereau PhD, CEO, and Luis Briseño-Roa PhD, CSO, as a spin-off at Institut Imagine, Paris, and already had very positive experience of Gyrolab xPlore and Gyros Protein Technologies. When the team at Medetia was first contacted by Olivier Gribouval, scientific equipment manager at Institut Imagine, about the need to scale up analysis to handle what would become over 34,000 samples, they immediately recommended transferring the immunoassays to Gyrolab xPand.
Paul Bastard‘s team was given access to two Gyrolab xPand systems, supported by Gyros Protein Technologies. Paul was really impressed with Gyrolab system. “For the first study I was using classical ELISA, which was a complete nightmare. Moving to Gyrolab system was a revelation. Suddenly we only had to prepare the plates and everything else was automated. After initial technical issues were solved in the first couple of weeks, analysis became routine and there was very little need to re-run samples”.
The pace was intense for the first two months when they processed 30,000 samples from the general population and involved measuring two autoantibodies against cytokines in 750 samples, or 1500 data points per working day from three 5-CD runs. “We were five PhD students taking turns to perform three runs per day, five days a week. Each run took seven hours, and we started the runs at around 7:30 am, 2 pm, and finished by starting an overnight run at 8–9 pm”. The pace slowed to two runs a day when most of the samples had been processed. They now perform two to three runs per week.
The autoantibody measurements were extremely reproducible, and the data proved to be very reliable. “The samples with a high titer according to the Gyrolab assays were well correlated with the data from other assays used to confirm the neutralization by autoantibodies. This gave us confidence in the Gyrolab assays.”
Luis Briseño-Roa from Medetia added, “Gyrolab system is so much more valuable thanks to the team around it. Other technologies we considered were quickly left behind by the rapid and effective support from Gyros Protein Technologies, especially when the throughput had to be quickly ramped up to meet the pressing timelines set by the ongoing pandemic.”
Autoantibodies are a major factor in age-related COVID-19 severity and morbidity
The above study included 3,595 patients hospitalized with critical COVID-19 pneumonia and a control group of 1,639 individuals with mild or no symptoms. High levels of type 1 IFN autoantibodies were found in 10.3% of critical patients or patients with severe COVID-19, and only 0.6% of the controls. This and other findings indicated that anti-type 1 IFN autoantibodies might be neutralizing type 1 IFNs, contributing to severe disease.
The researchers also used the Gyrolab assay to screen for IgG autoantibodies against IFN-α2 and IFN-ω in a group of 34,159 individuals from the general population who were free from SARS-CoV-2 infection. They found that autoantibodies capable of neutralizing high levels of cytokine are rare in the 18–69-year age group (0.18%) and then increase exponentially with age to 3.4% in the 80+ year-old group. A smaller study looking at autoantibodies capable of neutralizing lower cytokine levels showed similar trends. These patterns look remarkably like the infection fatality rates we looked at in the beginning (Figure 1).
Looking ahead
As the authors of the latest report (6) point out, their discoveries could lead to new ways of handling COVID-19:
- Test for autoantibodies, especially neutralizing antibodies, in patients infected with SARS-CoV-2 or in the general population.
- Preferentially vaccinate individuals with autoantibodies.
- Avoid live-attenuated vaccines when vaccinating individuals with autoantibodies.
- Ensure that unvaccinated individuals with autoantibodies are quickly managed.
- Screen blood products, especially plasma, for anti-IFN auto-Abs and exclude positive samples from donation.
Two recent publications have already underlined the value of these discoveries. In one study, patients with autoimmune polyendocrine syndrome type-1 (APS-1) with preexisting autoantibodies neutralizing type I IFNs have a very high risk of life-threatening COVID-19 pneumonia (7). In another study, a COVID-19 patient with incontentia pigmenti (IP), which results in reduced type 1 IFN levels and increased susceptibility to life-threatening COVID-19 pneumonia, was successfully treated with injections of IFN-ß1a that quickly resolved her symptoms (8).
It is work like this that puts into focus the enormous strides that are being made in handling infectious disease, and the value of technologies such as Gyrolab system that enable the high-throughput generation of reliable data.
References:
- Levin, AT et al. Assessing the age specificity of infection fatality rates for COVID-19: systematic review, meta-analysis, and public policy implications. Eur J Epidemiol. 2020 Dec;35(12):1123-1138. doi: 10.1007/s10654-020-00698-1.
- Bastard P, et al. Autoantibodies against type I IFNs in patients with life-threatening COVID-19. Science. 2020 Oct 23;370(6515). doi: 10.1126/science.abd4585. Epub 2020 Sep 24. PMID: 32972996.
- Asano, T et al. X-linked recessive TLR7 deficiency in ~1% of men under 60 years old with life-threatening COVID-19. Sci Immunol. 2021 Aug 19;6(62). doi: 10.1126/sciimmunol.abl4348.
- Kwon, D. Rogue antibodies linked to deaths from severe COVID. News in Focus, Nature, Vol 597, 9 September 2021.
- Viruses, microscopy and fast radio bursts: 10 remarkable discoveries from 2020. Highlights from News & Views published this year. Nature 588, 596-598 (2020). Doi: https://doi.org/10.1038/d41586-020-03514-8.
- Bastard, P et al. Autoantibodies neutralizing type I IFNs are present in ~ 4% of uninfected individuals over 70 years old and account for ~ 20% of COVID-19 deaths. Sci Immunol. Aug 19;6(62), 2021. doi: 10.1126/sciimmunol.abl4340.
- Bastard, P et al. Preexisting autoantibodies to type I IFNs underlie critical COVID-19 pneumonia in patients with APS-1. J Exp Med. 2021 Jul 5;218(7):e20210554. doi: 10.1084/jem.20210554.
- Bastard, P et al. Interferon-β therapy in a patient with Incontinentia Pigmenti and autoantibodies against Type I IFNs infected with SARS-CoV-2. J Clin Immunol. 2021 Jul;41(5):931-933. doi: 10.1007/s10875-021-01023-5. Epub 2021 Mar 25.